|
http://www.lipidlibrary.co.uk/Lipids/cmh/
MONOGLYCOSYLCERAMIDES (CEREBROSIDES)
Galactosylceramide (Galß1-1'Cer) is the principal glycosphingolipid in brain tissue, hence the trivial name "cerebroside",
which was first conferred on it in 1874, although it was much later before it was properly characterized. In fact, galactosylceramides
are found in all nervous tissues, but they can amount to 2% of the dry weight of grey matter and 12% of white matter.

Glucosylceramide (Glcß1-1'Cer) is also found at low levels in animal tissues, such as spleen and erythrocytes, as well
as in nervous tissues. It is a major constituent of skin lipids, and indeed the epidermal glucosylceramides are the source
of the unusual complex ceramides that are found in the stratum corneum (described on the ceramides page). Presumably, it functions as part of the water permeability barrier. In addition, higher than normal concentrations
of glycosphingolipids have been reported for the apical plasma membrane domain of epithelial cells from intestine and urinary
bladder. However, of greater importance than the natural occurrence of glucosylceramide per se is its role as the biosynthetic
precursor of lactosylceramide, and thence of the complex neutral oligoglycolipids and gangliosides.
| trop·ic [ tróppik ] |
noun (plural trop·ics) |
|
1. line of latitude: a line of latitude on
the Earth’s globe either 23º 26′ north of the equator tropic of Cancer or 23º 26′ south tropic of Capricorn
|
2. astronomy circle on the celestial sphere: either of two circles on the celestial sphere that have the same latitudes and mark the limits of the apparent
north-and-south movement of the Sun. The tropics lie in the same planes as the tropic of Cancer and the tropic of Capricorn.
|
plural noun trop·ics
or Trop·ics |
|
area between the tropics: the area between or near
the tropic of Cancer and the tropic of Capricorn
|
adjective |
|
http://encarta.msn.com/dictionary_1861722116/tropic.html
|
tropical: tropical
|
| [Early 16th century. Via Old French tropique from Latin tropicus from, ultimately, Greek tropē
“a turning,” from the ancient belief that the sun “turned back” at the tropics of Cancer and Capricorn.] |
Examples of stimuli are light, heat, food, or chemicals.
ampho- (ampho-) [Gr. amph[omacr] both] a prefix meaning both
internalization: to give a subjective character to; specifically : to incorporate (as values
or patterns of culture) within the self as conscious or subconscious guiding principles through learning or socialization
-
in·ter·nal·i·za·tion
cytokines
Non-antibody proteins secreted by inflammatory leukocytes and some non-leukocytic cells, that act as intercellular mediators. They differ from classical hormones in that they are produced by a number of tissue or cell types rather than by specialised glands. They generally act locally in a paracrine or autocrine rather than endocrine manner.
A substance that is produced by cells of the immune system and can affect the immune response. Cytokines can also be produced
in the laboratory by recombinant DNA technology and given to people to affect immune responses.
IL
abbr. for any of the subgroup of cytokines termed collectively interleukins because they were once thought to represent factors mediating communication between leukocytes. Individual factors are indicated
by numbers.
See: IL1 , IL2 , IL3 , IL4 , IL5 , IL6 , IL7 , IL8 , IL9 , IL10 , IL11 , IL12 , IL13 , IL14 , IL15 , IL16 , IL17 , IL17B , IL17C , IL17E , IL18 , IL19 , IL20 , IL21 , IL22 , IL23 , IL24 , IL25 , IL26 , IL27 , IL28A , IL28B , IL29 , IL30 .
Cytokines
The term cytokine, or immunocytokines , was used initially to separate a group of immunomodulatory proteins, called alsoimmunotransmitters , from otherGrowth factors that modulate the proliferation and bioactivities of non-immune cells. However, this terminology suggesting aclear-cut
distinction cannot be maintained and may not be meaningful altogether. Some cytokines are produced by a rather limited number
of different cell types while others are produced by almost the entire spectrum of known cell types.
The initial concept of "one producer cell -one cytokine -one target cell" has been falsified for practically every cytokine
investigated more closely. A definition of these factors on the basis of their producer or target cells is therefore also
problematic.
The same applies to classifications based upon identical or shared biological activities of cytokines especially with broad
definitions (see, for example: BCDF (B-cell differentiation factors ), BCGF (B-cell growth factors ), Motogenic cytokines , Chemotactic cytokines (see: Chemokines ), CSF (colony stimulating factors ), angiogenesis factors, or TRF (T-cell replacing factors )) ( for some personal views on aspects of nomenclature see also: Some personal remarks ).
Designations such as HBGF (heparin-binding growth factors ) take into account some biochemical shared by a variety of cytokines but are also problematic.
Today the term cytokine is used as a generic name for a diverse group of soluble proteins and peptides which act as humoral
regulators at nano- to picomolar concentrations and which, either under normal or pathological conditions, modulate the functional
activities of individual cells and tissues. These proteins also mediate interactions between cells directly and regulate processes
taking place in the extracellular environment (for some mechanistic concepts underlying cytokine actions see also: autocrine , paracrine , juxtacrine , retrocrine ). Many growth factors and cytokines act as cellular survival factors by preventing programmed cell death (see: Apoptosis ).
In many respects the biological activities of cytokines resemble those of classical hormones produced in specialized glandular
tissues. Some cytokines also behave like classical hormones in that they act at a systemic level, affecting, for example,
biological phenomena such as inflammation , systemic inflammatory response syndrome , and acute phase reaction , wound healing , and the neuroimmune network .
In general, cytokines act on a wider spectrum of target cells than hormones. Perhaps the major feature distinguishing cytokines
from mediators regarded generally as hormones is the fact that, unlike hormones, cytokines are not produced by specialized
cells which are organized in specialized glands, i. e. there is not a single organ source for these mediators. The fact that
cytokines are secreted proteins also means that the sites of their expression does not necessarily predict the sites at which
they exert their biological function.
Some cytokines have been found, upon determination of their primary structures, to be identical with classical enzymes
(see, for example: ADF (adult T-cell leukemia-derived factor ), nm23 , PD-ECGF (platelet-derived endothelial cell growth factor ), neuroleukin ). Cytokines normally do not possess enzymatic activities although there is a growing list of exceptions.
The biological activities of cytokines can be measured by a variety of Bioassays employing, among other things, Factor-dependent cell lines , or by other assays using, for example, antibodies (see also: Cytokine assays , WHO cytokine standardization ). RT-PCR quantitation of cytokines employs modern techniques of molecular biology and detects the presence of mRNA encoding specific cytokines.
In the more restricted sense cytokines comprise Interleukins , initially thought to be produced exclusively by leukocytes, Lymphokines , initially thought to be produced exclusively by lymphocytes, Monokines , initially thought to be produced exclusively by monocytes, interferons (see: IFN ), initially thought to be involved in antiviral responses, colony stimulating factors (see: CSF ), initially thought to support the growth of cells in semi-solid media (see also: Colony formation assay ), Chemokines , thought to be involved in Chemotaxis , and a variety of other proteins.
The termType-1 cytokines refers to cytokines produced by Th1 T-helper cells whileType-2 cytokines are those produced by Th2 T-helper cells. Type-1 cytokines include IL2 , IFN-gamma , IL12 and TNF-beta , while Type-2 cytokines include IL4 , IL5 , IL6 , IL10 , and IL13 .
It has been suggested that the generic termPeptide regulatory factors ( abbr. PRF ) be used for all these factors to avoid the general difficulties with the nomenclature (see also: Some personal remarks ). This term has the advantage that it includes also a number of low molecular mass peptides which are generally not regarded
as cytokines although they have many activities of cytokines. Some of these low molecular weight proteins and peptides have
been referred to asMinicytokines .
A comparison of sequences demonstrates that nonhuman primate cytokines are closely related. For example, IL1-alpha , IL1-beta , IL2 , IL4 , IL5 , IL6 , IL8 , IL10 , IL12 , IL15 , IFN-alpha , IFN-gamma , and TNF-alpha share 93 to 99 percent homology at the nucleic acid and protein level with the human orthologous sequences (Villinger et al).
Cytokines from other species are frequently detected by virtue of sequence homologies.
Most cytokines are unrelated in terms of sequence although some can be grouped into families (see: Gene family ; see also: Cytokine receptor families ) or are classified into categories according to the types of secondary and tertiary structure. For example, IL6 , IL11 , CNTF , LIF , OSM , Epo , G-CSF , GH , PRL , IL10 , IFN-alpha , IFN-beta form long chain 4 helix bundles. IL2 , IL4 , IL7 , IL9 , IL13 , IL3 , IL5 , GM-CSF , M-CSF , SCF , IFN-gamma form short chain 4 helix bundles. So-called beta-trefoil structures are formed by IL1-alpha , IL1-beta , aFGF , bFGF , int-2 , KGF . EGF-like antiparallel beta-sheets are formed by EGF , TGF-alpha , Betacellulin , SCDGF , Amphiregulin , HB-EGF . For other aspects of biochemistry see also: Recombinant cytokines , Muteins , Peptide mimetics .
Most cytokines are glycoproteins which are secreted by cells using classical secretory pathways (see also: signal sequence ). Many genes encoding cytokines can give rise to a variety of variant forms of cytokines by means of alternative splicing,
yielding molecules with slightly different but biologically significant bioactivities. In many cases the expression patterns
of different forms of cytokines or of members of a cytokine family are overlapping only partially, suggesting a specific role
for each factor.
Membrane-bound forms have been described also for many cytokines, and some may be associated also with the extracellular matrix . It is likely that the switching between soluble and membrane forms of cytokines is an important regulatory event (see also:
Autocrine , paracrine , juxtacrine , retrocrine ). In some cases membrane forms of a cytokine have been found to be indispensable for normal development, with soluble forms
being unable to entirely substitute for them.
Most cytokines are generally not stored inside cells (exceptions are, for example TGF-beta and PDGF which are stored in platelets). The expression of most cytokines is regulated tightly at practically all levels: these factors
are usually produced only by cells after cell activation in response to an induction signal. Expression can be regulated at the level of transcription, translation, and protein synthesis
(see also: gene expression ; ARE (AU-rich element )). Normally, cytokines are expressed transiently only but constitutive expression has been observed also. The expression
of many cytokines also seems to be regulated differentially, depending on cell type and developmental age. Secretion or release
from producer cells is a regulated process. Once released, their behaviour in the circulation may be regulated by soluble
receptors and specific or unspecific binding proteins. Regulation also is at work at the receptor level on target cells and
at the level of signaling pathways governing alterations in the behaviour of responder cells.
Most cytokines were detected initially in functional tests in vitro as biochemically undefined activities or as distinct
factors with distinct biological activities. This also explains, at least in part, the plethora of different names for some
of the cytokines. In many instances these activities were named after a particular biological activity observed in an in vitro
assay (see also: Bioassays and Cytokine assays for alternatives) or after cells that were found to elaborate these factors (for techniques allowing identification of cytokine
genes, cytokine receptor genes, and other relevant genes without prior knowledge of their activities see: Gene library ). One should be aware of the fact that at this moment in time the relevance of many in vitro activities of cytokines to their
endogenous functions within an intact organism is not clearly defined.
Almost all cytokines are pleiotropic effectors showing multiple biological activities. In addition, multiple cytokines
often have overlapping activities and a single cell frequently interacts with multiple cytokines with seemingly identical
responses (cross-talk). One of the consequences of this functional overlap is the observation that one factor may frequently
functionally replace another factor altogether or at least partially compensate for the lack of another factor. Since most
cytokines have ubiquitous biological activities, their physiologic significance as normal regulators of physiology is often
difficult to assess.
Studies of gene functions in experimental transgenic animals in which a cytokine gene has been functionally inactivated by gene targeting (see also: Knock-out ) are of particular importance in research on cytokines because, unlike in vitro studies, they provide information about the
true in vivo functions of a given cytokine by highlighting the effects of their absence. In many instances these studies have
shown that null mutations of particular cytokine genes do not have the effects in vivo expected from their activities in vitro. If information about
loss-of-function studies is available for a given cytokine or its receptor and if I had time to add the information it can
be found as a special subentry (Transgenic/Knock-out/Antisense studies) for each particular cytokine.
Many cytokines show stimulating or inhibitory activities and may synergise or antagonize also the actions of other factors.
A single cytokine may elicit reactions also under certain circumstances which are the reverse of those shown under other circumstances.
The type, the duration, and also the extent of cellular activities induced by a particular cytokine can be influenced considerably
by the micro-environment of a cell, depending, for example, on the growth state of the cells (sparse or confluent), the type
of neighboring cells, cytokine concentrations, the combination of other cytokines present at the same time, and even on the
temporal sequence of several cytokines acting on the same cell. Under such circumstances combinatorial effects thus allow
a single cytokine to transmit diverse signals to different subsets of cells.
The fact that every cell type may have different responses to the same growth factor can be explained, at least in part,
by different spectrums of genes expressed in these cells and the availability and levels of various transcription factors that drive Gene expression . The responses elicited by cytokines are therefore contextual and the "informational content", i. e. the intrinsic activities
of a given cytokine may vary with conditions. Although a variety of cytokines are known to share at least some biological
effects the observations that single cells usually show different patterns of gene expression in response to different cytokines can be taken as evidence for the existence of cytokine receptor-specific signal transduction
pathways. Shared and different transcriptional activators that transduce a signal from a cytokine receptor to a transcription
regulatory element of DNA are involved in these processes (for some examples see: STAT proteins , Janus kinases , IRS ).
It has been observed, for example, that bFGF is a strong mitogen for fibroblasts at low concentrations and a chemoattractant at high concentrations (see also: Chemotaxis ). bFGF has been shown also to be a biphasic regulator of human hepatoblastoma-derived HepG2 cells, depending upon concentration. The interferon IFN-gamma can stimulate the proliferation of B-cells prestimulated with Anti-IgM, and inhibits the activities of the same cells induced
by IL4 . On the other hand, IL4 activates B-cells and promotes their proliferation while inhibiting the effects induced by IL2 in the same cells. The activity of at least two cytokines (IL1-alpha and IL1-beta ) is regulated by an endogenous receptor antagonist, the IL1 receptor antagonist (see: IL1ra ). Several cytokines, including TNF , IFN-gamma , IL2 and IL4 , are inhibited by soluble receptors (see also: Receptor shedding , Cytokine inhibitors , retrocrine ). Several cytokines, including IL10 and TGF-beta , act to inhibit other cytokines.
The processes responsible for the regulation of cytokines are not well understood. Cells utilize distinct biochemical pathways
converging on mediator release and these can be probed, among other things, by employing a variety of substances mimicking
or inhibiting the actions of cytokines (see, for example: Bryostatins , Calcium ionophore , Genistein , H8 , Herbimycin A , K-252a , Lavendustin A , Phorbol esters , Okadaic acid , Staurosporine , Suramin , Tyrphostins , Vanadate ).
Frequently one observes a hierarchical order of cytokine actions with some early Cytokines preactivating cells so that
they then can respond to late-acting cytokines (see also: Cell activation ). Many cytokines induce the synthesis of novel gene products once they have bound to their respective receptors (see: ERG , Early response gene ). Some of the novel products are themselves cytokines (see: Chemokines , for example). In addition, there are a variety of biological response modifiers that function as Anti-cytokines .
Cytokine mediators can be transported quickly to remote areas of a multicellular organism. They can address multiple target
cells and can be degraded quickly. Concentration gradients can be used to elicit specific responses. These possibilities by
far exceed the possibilities provided by mere cell-to-cell contacts within a multicellular organism. It can be assumed that
cytokines play a pivotal role in all sorts of cell-to-cell communication processes although many of the mechanisms of their
actions have not yet been elucidated in full detail.
A close examination of the physiological and pathological effects of the regulated or deregulated (see: Transgenic animals) expression of cytokines in complex organisms has shown that these mediators are involved in virtually all general
systemic reactions of an organism (see also: CytokineTopics ), including such important processes as the regulation of immune responses (see, for example: BCDF , B-cell growth and differentiation factors; BCGF , B-cell growth factors ; TRF , T-cell replacing factors ; Isotype switching ), inflammatory processes (see: Inflammation ), hematopoiesis (see also: Hematopoietins ), and wound healing .
Cytokines are important mediators involved in embryogenesis and organ development (see also: Angiogenesis ) and their activities in these processes may differ from those observed postnatally. In addition they play a key role in
neuroimmunological, neuroendocrinological, and neuroregulatory processes (see: Neuroimmune network ). Cytokines are important positive or negative regulators of mitosis (see also: Cell cycle ), differentiation, migration (see also: Chemotaxis , Chemokines ), cell survival and cell death (see also: Apoptosis ), and transformation (see also: Oncogene ). It has been shown that a number of viral infectious agents exploit the cytokine repertoire of organisms to evade immune
responses of the host. Virus-encoded factors (see also: Virulence Factors MiniCOPE Dictionary .) appear to affect the activities of cytokines in at least four different ways: by inhibiting the synthesis and release of
cytokines from infected cells; by interfering with the interaction between cytokines and their receptors; by inhibiting signal
transmission pathways of cytokines; and by synthesizing virus-encoded cytokines that antagonize the effects of host cytokines
mediating antiviral processes (see: Viroceptor , Virokine ). Bacteria and other micro-organisms also appear to produce cytokine-like substances which they utilize to subvert host responses
(see: Bacteriokine , Microkine ).
Cytokines themselves rarely are related closely among each other in terms of primary sequences. Some appear to have some
common three-dimensional features and some of them can be grouped into families. For example, the TNF ligand superfamily members (with the exception of LT-alpha ) are type-2 membrane glycoproteins (N-terminus inside) with homology to TNF in the extracellular domain (overall homologies, 20 percent. The HBNF family includes members of the group of fibroblast growth factors . Another group of diverse factors with conserved sequence features are the Chemokines . The analysis of crystal structures of several cytokines with very little sequence homology has revealed a common overall
topology that is not deducible from sequence comparisons (see: Cystine knot growth factor family ).
The biological activities of cytokines are mediated by specific membrane receptors which can be expressed on virtually
all cell types known. Their expression is also subject to several regulatory mechanisms (see: Receptor transmodulation ) although some receptors are expressed also constitutively.
Cytokine receptor proteins have been shown to share a number of characteristics. Many receptors are members of cytokine receptor families . Many receptors are multi-subunit structures that bind ligands and at the same time possess functions as signal transducers
due to their intrinsic tyrosine kinase activity (see also: Autophosphorylation ; see also: PTK ; protein tyrosine kinase ). Many receptors often share common signal transducing receptor components in the same family (see also: Cytokine receptor families ), which explains, at least in part, the functional redundancy of cytokines. It is the cross-communication between different
signaling systems that eventually allows the integration of a great diversity of stimuli, which a cell can be subjected to
under varying physiological situations. This and the ubiquitous cellular distribution of certain cytokine receptors has hampered
attempts to define critical responsive cell populations and the physiologically important cell-specific functions of cytokines
in vivo. Many receptors are associated with special signal transducing proteins in the interior of the cell (see, for example
Janus kinases , STAT proteins ). Some receptors may bind more than one cytokine. Several cytokine receptors have been shown to be converted into soluble
binding proteins that regulate ligand access to the cell by specific proteolytic cleavage of receptor ectodomains.
The many specific activities of individual cytokines have been the basis for current concepts of therapeutical intervention,
in particular of the treatment of hematopoietic malfunctions and tumor therapy. Applications involve the support of chemo-
and radiotherapy, bone marrow transplantation, and general immunostimulation (see also: Adoptive immunotherapy , LAK cells , TIL , Cytokine gene transfer , Cytokine fusion toxins ).
Although some recombinant cytokines are now in clinical use, and and attempts are made to develop hybrid molecules from known cytokines (see: Muteins ) which possess the advantages of the respective factors, but not their disadvantages, one must be aware of the fact that
current knowledge is still limited. Cytokines are powerful two-edged weapons that can trigger a cascade of reactions, and
may show activities that often go beyond the single highly specific property which it is hoped they possess. New factors are
being discovered constantly and they extend our knowledge about the Cytokine network .
Nevertheless it can be stated that our new (and growing) understanding of the biological mechanisms governing cytokine
actions are an important contribution to medical knowledge. The biochemistry and molecular biology of cytokine actions explain
some well-known and sometimes also some of the more obscure clinical aspects of diseases. Knowledge that cytokines create
regulatory hierarchies and provide independent and/or interrelated regulatory mechanisms that can confer distinct and interactive
developmental functions lays a solid, albeit rather complicated foundation, for current and future clinical experiences.
date of last revision: March 2003
References: Balkwill FR and Balkwill F ( editors) Cytokine Molecular Biology: A Practical Approach, Oxford University
Press (2000); Balkwill FR ( editor) Cytokine Cell Biology: A Practical Approach, 3rd edition, Oxford University Press
(2001); Callard RE ( editor) Cytokines and B lymphocytes. Academic Press, London, 1991; Dinarello CA et al (
editors) The physiological and pathological effects of cytokines. Liss, New York, 1990; Habenicht A ( editor) Growth
factors, differentiation factors, and cytokines, Springer, Berlin 1990; Nilsen-Hamilton M ( editor) Growth factors
and development. ( Current Topics in Developmental Biology, Vol. 24, Academic Press 1990; Oppenheim J Immunophysiology:
The Role of Cells and Cytokines in Immunity and Inflammation, Oxford University Press (1991); Oppenheim J et al ( editors)
Cytokine Reference: A Compendium of Cytokines and Other Mediators of Host Defense, Academic Press (2000); Ransohoff RM
( Editor), Cytokines and the CNS, CRC Press (1996); Villinger F et al Comparative sequence analysis of cytokine genes
from human and nonhuman primates. Journal of Immunology 155(8): 3946-3954 (1995); Westwick SL et al ( eds) Chemotactic
cytokines. Plenum Press, New York 1991)
Muteins
[ mutant proteins] A general term describing mutated recombinant proteins. These proteins usually carry single or multiple
amino acid substitutions. These proteins are derived frequently from cloned genes that have been subjected to site-directed
or random mutagenesis, or from completely synthetic genes.
High-resolution mutational and structural analyses have revealed a great deal about the molecular basis for the actions
of cytokines . From these studies it has been possible to engineer homologues of cytokines with altered biological properties. Cytokines with greater specific activity in cell proliferation assays (see: Bioassays ), enhanced antitumor activities, resistance to proteolytic cleavage, or improved pharmacokinetic properties are superior
to the parent proteins and are particularly useful, therefore, in clinical applications.
For specific examples of cytokine muteins see: CS23 (a mutein of bFGF ), KW-2228 (a mutated form of G-CSF ), Daniplestim (a mutant IL3 ), pIXY321 (a fusion protein derived from the coding sequences of GM-CSF and IL3 ), Consensus interferon (see also: Recombinant cytokines ), Leridistim , Myelopoietin , TNF-alpha muteins . For a human recombinant fusion protein consisting of IL6 and IL2 see: CH925 . Mutations at Leu25 of IL8 have been shown to create a factor with altered chemokine receptor selectivity and novel chemokine-like properties. Protease-resistant
muteins have been described for IGF-1 by Bryant et al. For muteins comprising agonists that combine activites binding to the human fetal liver tyrosine kinase flt-3 and the G-CSF receptor see: progenipoietin . For chimeric dual agonist of the cellular mpl gene, the receptor for thrombopoietin , and the human IL3 receptor see: Promegapoietin .
Methods of genetic engineering have been used also to produce so-calleddesigner cytokines with improved or enhanced bioactivities. One example is the construction of a fusion protein consisting of the amino acid
chains of IL6 and a soluble IL6 receptor in which the two moieties are linked by a flexible peptide chain (see: H-IL6 ). Another example is the construction of a bioactive murine and human fusion protein combining two different subunits of
IL12 in a single molecules. Anderson et al describe the construction of Flexi 12 , a single chain protein retaining all of the biological characteristics of the dimeric recombinant IL12 .
A third example is Pan-Neurotrophin-1 developed by Ilag et al, which combines structural elements of NGF , BDNF , and NT-3 (see also: Neurotrophins ).
Ye et al have constructed a fusion protein consisting of GM-CSF and MCAF . The recombinant human fusion protein can sustain the growth of GM-CSF dependent cell line TF-1 and is chemotactic for monocytes. In vitro the fusion protein activates monocytes to inhibit the growth of several human tumor
cell lines. The fusion protein is more effective in inducing cytotoxicity of monocytes than GM-CSF or MCAF alone or than a combination of GM-CSF and MCAF . The fusion protein also completely suppresses tumor formation in nude mice.
Difalco and Congote describe the preparation of a recombinant chimaera of IGF-2 and human IL3 and the production of this protein using the Baculovirus expression system . The chimaeric protein displays a high proliferative potency for haemopoietic cells. The chimaera has a significantly higher
mitogenic activity than IL3 in cell cultures of the human haemopoietic cell line TF-1 . It was also able to stimulate thymidine incorporation in IGF-2 dependent bovine fetal erythroid cells. Subcutaneous injection of a similar protein containing mouse IL3 sequences in normal C57BL/6 mice was shown to result in a significant increase of the number of spleen stem cells producing
macroscopic hematopoietic colonies (see also: Colony formation assay ).
FP6 is a fusion protein consisting of the soluble form of the IL6 receptor lacking the transmembrane domain and cytoplasmic regions and IL6 .
Grazi-Cusi and Ferrero have produced a series of artificial cytokines derived from sequences of human IL6 and G-CSF , which they termedHarlequin molecules . Some of these chimeric molecules maintain the activity of either IL6 or G-CSF and at least one (Harlequin-11) has both biological activities in various assay systems. One chimeric protein appears to compete
for the activity of the intact cytokine.
For other therapeutically valuable hybrid constructs see also: Cytokine fusion toxins , Antibody fusion proteins . For peptides showing the same activities as the cognate ligands of a cytokine receptor see also: Peptide mimetics . For a naturally occurring hybrid see: hybrid cytokine .
date of last revision: March 2003
References: Anderson R et al Construction and biological characterization of an interleukin 12 fusion protein (flexi
12): delivery to acute myeloid leukemic blasts using adeno associated virus. Human Gene Therapy 8(9): 1125-1135 (1997); Bryant
KJ et al Design and characterisation of long-R3-insulin-like growth factor-I muteins which show resistance to pepsin digestion.
Growth Factors 13(3-4): 261-272 (1996); Difalco MR and Congote LF Preparation of a recombinant chimaera of insulin-like
growth factor II and interleukin 3 with high proliferative potency for haemopoietic cells. Biochemical Journal 326(2): 407-413
(1997); Fischer M et al A bioactive designer cytokine for human hematopoietic progenitor cell expansion. Nature Biotechnology
15: 142-145 (1997); Grazi-Cusi M ; Ferrero D Harlequin granulocyte-colony stimulating factor interleukin 6 molecules
with bifunctional and antagonistic activities. Immunotechnology 3(1): 61-69 (1997); Ilag LL et al Pan-neurotrophin
1: a genetically engineered neurotrophic factor displaying multiple specificities in peripheral neurons in vitro and in vivo.
Proceedings of the National Academy of Science (USA) 92(2): 607-611 (1995); Lustinarasimhan M et al A molecular switch
of chemokine receptor selectivity: chemical modification of the interleukin 8 leu(25)cys mutant. Journal of Biological Chemistry
271(6): 3148-3153 (1996); Ibanez CF et al An extended surface of binding to Trk tyrosine kinase receptors in NGF and
BDNF allows the engineering of a multifunctional pan-neurotrophin EMBO Journal 12: 2281-2293 (1993); Lieschke GJ et al Bioactive murine and human interleukin-12 fusion proteins which retain antitumor
activity in vivo. Nature Biotechnology 15: 35-40 (1997); Ye QN et al Construction of a recombinant human GM CSF/MCAF
fusion protein and study on its in vitro and in vivo antitumour effects. Science in China Series C Life Sciences 40(1): 18-26
(1997)
Knock-out
A general term (sometimes abbr. GKO ,gene knock-out ) relating to artificially generatednull mutations ( referred to also asloss-of-function mutations ) of a gene.
Such mutants, which are devoid, for example, of a particular cytokine or receptor function, are created by a process calledhomologous recombination ,targeted deletion , ortargeted disruption . It essentially involves the inactivation of an endogenous fully functional gene by insertion of cloned sequences.
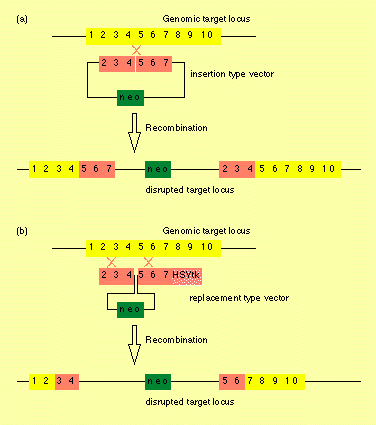 |
| Principle of gene targeting involving the use of insertion or replacement type vectors.
( a) Use of insertion type vectors involves a single cross-over between genomic target sequences and homologous sequences at either end of the targeting vector. The neomycin resistance gene contained within the vector serves as a positive
selectable marker.
(b ) Gene targeting using replacement type vectors requires two cross-over events. The positive selection marker (neo) is retained while the negative
selectable marker (HSV thymidine kinase) is lost. The advantage of this system is the fact that cells harboring randomly and
unspecifically integrated gene constructs still carry the thymidine kinase gene. These cells can be eliminated selectively
by using thymidine kinase as a selective marker. One disadvantage of the system may be the time required to handle ES cells in vitro. These cells have a certain tendency to differentiate and this can favor the subsequent generation of genetic mosaics
rather than the desired germ line integration when the cells are used to create transgenic animals. |
In mice the process involves manipulation of ES cells which are used subsequently to regenerate transgenic animals carrying the genetic defect. The technique is applicable, however, to virtually all biological cell systems employed
in molecular biology and cell biology.
Another way to inactivate specifically a given (cytokine) gene is the use of antisense RNA. Special techniques such as genetic ablation allow generation of animals in which only one or several cell types have been modified genetically so that they are subsequently
lacking completely.
Since most cytokines have ubiquitous biological activities, their physiologic significance as normal regulators of physiology is often difficult
to assess (see also: Cytokine network ). Studies of gene functions and biological processes in knock-out mice and other organisms have been of particular importance
in cytokine research. Unlike in vitro studies, this approach provides information about the true physiological functions of
a given cytokine in vivo by studying the biological consequences of its absence. The use of cell lines lacking expression
of a particular protein is instrumental also in studying potential and proposed target proteins and thus is critical for evaluating
signalling mechanisms. This is because cells not expressing a particular protein may be found to be responsive or unresponsive
to another protein under study.
In some instances studies with cytokine knock-out mice have revealed either that these mice develop normally and/or do
not show the pronounced effects on the immune system, hematopoiesis , or other organ systems one would have expected from the known in vitro activities of the cytokines (for a good example see: IL2 , IL4 ).
If information about knock-out studies is available for cytokines or their receptors it can be found in the COPE encyclopaedia of cytokines as a special subentry (Transgenic/Knock-out/Antisense studies) for each particular cytokine.
IFN
abbr. for interferons . By definition interferons are proteins that, at least in homologous cells, elicit a virus-unspecific antiviral activity. This activity requires new synthesis of RNA and proteins and is not observed
in the presence of suitable RNA and protein synthesis inhibitors.
Apart from their antiviral activities interferons also possess antiproliferative and immunomodulating activities and influence the metabolism, growth and differentiation of
cells in many different ways.
The three main human interferons are known asIFN-alpha ,IFN-beta andIFN-gamma . IFN-alpha and IFN-beta as well as IFN-delta , IFN-omega , and IFN-tau are called alsoType-1 interferon . IFN-gamma has been designatedType-2 interferon . Another protein, called originally IFN-beta-2 is not an interferon but is identical with IL6 . IFN-omega , IFN-tau (see: TP-1 [trophoblast protein-1 ]), and Limitin are other factors related to interferons .
Some older names of interferons such asleukocyte interferon ( IFN-alpha ), fibroblast interferon ( IFN-beta ) andimmune interferon ( IFN-gamma ) are still in use. These names are derived from the main producers and from the typical bioactivity and still reflect the
concept of a typical producer cell (see also: Cytokines ). This concept has been discarded now because it is known that a plethora of different cell types are capable of producing
interferons .
Interferons are a heterogeneous group of proteins with some similar biological activities that are distinguished from each other by many
different physical and immunochemical properties. They are also encoded by different structural genes. Most interferons are multifunctional proteins with bioactivities that are strictly species-specific (see: TP-1 , trophoblast protein-1 , for an exception). These substances are synthesized following the activation of the immune system. The human interferons IFN-beta and IFN-gamma are encoded by two different single genes while human IFN-alpha constitutes a family of at least 23 different genes.
Although there are some indications of constitutive interferon synthesis by some cell types interferons are generally inducible proteins. Their synthesis is induced by many different physiological and non-physiological inducers
including, among others, nucleic acids, synthetic oligonucleotides (polyIC), pyran copolymers, bacterial lipopolysaccharides , and a number of low-molecular weight compounds.
The production of interferons in an organism may be localized or, depending on the strength of the inducing stimulus, systemic, eventually culminating in
an interferonaemia. The detection of interferons in the circulation is usually an indication of some pathological state.
Interferons are mainly known for their antiviral activities against a wide spectrum of viruses. Interferons are synthesized, for example, by virus-infected cells and protect other, non-infected but virus-sensitive cells against infection
for some time. In addition interferons are also known to have protective effects against some non-viral pathogens.
The antiviral activities of interferons are used also to detect these proteins in Bioassays . Interferons possess very high specific activities. It is on the order of 10**9 units/mg of protein for IFN-alpha and IFN-beta . One unit is defined as the amount of interferon that reduced virus multiplication by 50 percent under standardized conditions.
Interferons are also potent immunomodulators. They can promote or inhibit the synthesis of antibodies by activated B-cells and also activate
macrophages, natural killer cells, and T-cells. Interferons mainly influence early unspecific immune response processes mediated predominantly by monocytes/macrophages. Among other things
interferons increase antigen and receptor expression in effector cells, induce the expression of new genes, inhibit the expression of
some genes, and also prolong phases of the cell cycle . Interferons also influence differentiation and developmental processes which is exemplified by their effects on the maturation of immature
muscle cells, the induction of globin genes, the methylation of tRNA, and the expression of carcinoembryonic antigen on tumor
cells.
Interferons also possess direct antiproliferative activities and are cytostatic or cytotoxic for a number of different tumor cell types.
These activities are partly due to complex interactions with other growth factors and their receptors the expression of which may be stimulated or inhibited by interferons . Many growth factors are capable also of inducing the synthesis of interferons .
Hormone-like activities of interferons are observed in cells of the central nervous and the neuroendocrine system. Interferons modulate central opioid functions and can induce alterations in sleep and behavioral pattern s at higher doses administered systemically. IFN-alpha significantly increases the firing rate of spontaneous active cells in the hippocampus and the somatosensoric cortex.
IFN-alpha and IFN-beta are thought to bind to the same receptor on the cell surface of target cells. This receptor differs from that of IFN-gamma . Binding of an interferon to its receptor induces the expression of a number of new proteins. Some genes are specifically
induced either by IFN-alpha and IFN-beta or by IFN-gamma . Some of the newly formed proteins are nucleases, synthases and protein kinases that influence protein biosynthesis and are,
at least in part, also responsible for some biological effects, for example, antiviral activities.
The induced protein kinase inactivates the eukaryotic initiation factor eIF-2 and thus inhibits the synthesis of new viral
proteins. The expression of 2',5'-polyadenylate synthetase which is induced also by interferons , leads to products that themselves activate endonucleases that in turn degrade viral mRNAs. Interferons do not show antiviral activities in undifferentiated embryonic carcinoma cells (EC cells ) that do not express certain endonucleases.
Of major clinical interest are the antiviral and growth-inhibitory activities of interferons while their use as immunoregulators is still in its infancy. All interferons show a similar spectrum of side effects with adverse effects observable more or less in all organs. Some of the side effects
may actually be dose-limiting although all symptoms appear to be readily reversible if treatment is discontinued.
date of last revision: January 2002
References: Borden EC Interferons: Pleiotropic cellular modulators. Clin. Immunology Immunopathol. 62: S18-S24 (1992);
Byrne GI and Turco J Interferon and non-viral pathogens. Marcel Dekker, New York, 1988; DeMaeyer E et al Immuno-modulating
properties of interferons. Philosophical Transactions of the Royal Society London 299: 77-90 (1982); DeMaeyer E and DeMaeyer-Guignard
J Interferons and other regulatory cytokines. Journal of Wiley, New York, 1988; Fent K and Zbinden G Toxicity of
interferon and interleukin Trends in Pharmacological Sciences 8: 100-5 (1987); Garbe C and Krasagakis K Effects of interferons and cytokines on melanoma cells. Journal of Investigative
Dermatology 100: 239s-44s (1993); Kardamakis D Interferons in the treatment of malignancies. In vivo 5: 589-98 (1991);
Kerr IM and Stark GR The antiviral effects of the interferons and their inhibition. Journal of Interferon Research
12: 237-40 (1992); Murray HW et al The interferons, macrophage activation, and host defense against nonviral pathogens.
Journal of Interferon Research 12: 319-22 (1992); Pestka S et al Interferons and their actions. Annual Review of Biochemistry
56: 727-77 (1987); Rubinstein M Multiple interferon subtypes: The phenomenon and its relevance. Journal of Interferon
Research 11: ( Suppl. Jan) 3-9 (1991); Staeheli P Interferon-induced proteins and the antiviral state. Advances in
Virus Research 38: 147-200 (1990); Tanaka N and Taniguchi T Cytokine gene regulation: regulatory cis elements and DNA
binding factors involved in the interferon system. Advances in Immunology 52: 263-81 (1992); Williams BRG Transcriptional
regulation of interferon-stimulated genes. European Journal of Biochemistry 200: 1-11 (1991)
http://www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=rv.section.288
retro virus:
The viral genes gag, pro, pol, and env (Table 1) occupy the body of the DNA. Indeed, for many retroviruses, these are the only genes. gag encodes the internal structural
protein of the virus (Gag protein, from the original name "group-specific anti gen," reflecting what
were believed to be the antigenic properties of this protein). Gag is proteolytically processed into the mature proteins MA
(matrix), CA (capsid), NC (nucleocapsid), and sometimes others, of uncertain function, that are designated by numbers.
pol encodes the enzymes reverse transcriptase (RT), which contains both DNA polymerase and associated RNase H activities,
and integrase (IN), which mediates replication of the genome. pro encodes the viral protease (PR), which acts late
in assembly of the viral particle to process proteolytically the proteins encoded by gag, pro, and pol,
and in some cases also env (Chapter 7). env encodes the surface (SU) glycoprotein and the transmembrane (TM) protein of the virion, which form a complex
that interacts specifically with cellular receptor proteins. This interaction leads ultimately to fusion of the viral membrane
with the cell membrane (Chapter 3). A few retroviral groups also contain another gene, called dut, which encodes a deoxyuridine triphosphatase (dUTPase
or DU). Unlike all other genes, dut is found in differing locations, being translated in the pro reading frame
in the B- and D-type viruses, and in the pol frame in the nonprimate lentiviruses.
In addition to gag, pro, pol, and dut, those retroviruses classified as complex the lentivirus genus, the spumavirus genus, the HTLV/bovine leukemia virus (BLV) genus, and a newly characterized fish virus
genus the lentivirus genus, the spumavirus genus, the HTLV/bovine leukemia virus (BLV) genus, and a newly characterized fish virus
genus also contain "accessory" genes. Accessory genes regulate and coordinate viral gene expression, and some also have other ancillary
roles. These genes are located between pol and env, just downstream from env including the U3 region
of the LTR, or overlapping portions of env and each other. also contain "accessory" genes. Accessory genes regulate and coordinate viral gene expression, and some also have other ancillary
roles. These genes are located between pol and env, just downstream from env including the U3 region
of the LTR, or overlapping portions of env and each other.
Some retroviruses carry genes of a different class: the oncogenes, or onc genes. Many retroviruses hereafter referred to as "transforming" viruses hereafter referred to as "transforming" viruses were first identified by their ability to rapidly cause tumors in animals and oncogenically transform cells in culture. Transformation
of cultured cells invariably was traced to host-derived sequences that the virus had acquired (Chapter 10). With the exception of some strains of RSV, retroviruses that carry oncogenes are defective, having suffered variable deletions
of one or more of the viral genes needed for replication during or after the acquisition event. As a consequence, many retroviral
oncogenes are expressed as Gag-Onc fusion proteins, with part or most of gag being deleted. There are also numerous
examples in which the oncogene replaces env or is positioned elsewhere in the genome. Retroviruses with such rearrangements
are defective for replication on their own and can replicate only if the cell is also infected with a nondefective virus,
usually called a helper virus. In the nondefective strains of RSV, the v-src oncogene is freestanding downstream from
env and is expressed from a separately spliced mRNA (Chapter 6). An overview of the organization of genes in a few prototypic retroviruses is presented in Figure 5. were first identified by their ability to rapidly cause tumors in animals and oncogenically transform cells in culture. Transformation
of cultured cells invariably was traced to host-derived sequences that the virus had acquired (Chapter 10). With the exception of some strains of RSV, retroviruses that carry oncogenes are defective, having suffered variable deletions
of one or more of the viral genes needed for replication during or after the acquisition event. As a consequence, many retroviral
oncogenes are expressed as Gag-Onc fusion proteins, with part or most of gag being deleted. There are also numerous
examples in which the oncogene replaces env or is positioned elsewhere in the genome. Retroviruses with such rearrangements
are defective for replication on their own and can replicate only if the cell is also infected with a nondefective virus,
usually called a helper virus. In the nondefective strains of RSV, the v-src oncogene is freestanding downstream from
env and is expressed from a separately spliced mRNA (Chapter 6). An overview of the organization of genes in a few prototypic retroviruses is presented in Figure 5.
The genes in the viral DNA are bracketed by the long terminal repeats (LTRs), identical sequences that can be divided into
three elements. U3 is derived from the sequence unique to the 3![[prime prime or minute]](http://www.ncbi.nlm.nih.gov/books/bookres.fcgi/system_default/prime.gif) end of the RNA, R is derived from a sequence repeated at both ends of the RNA, and U5 is derived from the sequence unique
to the 5 end of the RNA, R is derived from a sequence repeated at both ends of the RNA, and U5 is derived from the sequence unique
to the 5![[prime prime or minute]](http://www.ncbi.nlm.nih.gov/books/bookres.fcgi/system_default/prime.gif) end of the RNA. The genesis of the LTR elements lies in the process of reverse transcription, whereby the enzyme "jumps" from
one end of the template to the other (Chapter 4). The sizes of these three elements vary considerably among different retroviruses, with U3 typically ranging from several
hundred nucleotides to more than a thousand nucleotides, R from a dozen to more than a hundred nucleotides, and U5 from about
one to two hundred nucleotides (Table 2). From the definition of U3, R, and U5, it follows that the site of transcription initiation is at the boundary between U3
and R, and the site of poly(A) addition is at the boundary between R and U5, as shown. The other boundaries of U3 and U5 are
determined by the sites of initiation of plus- and minus- strand DNA synthesis. U3 contains most of the transcriptional control
elements of the provirus, which include the promoter proper, and multiple enhancer sequences responsive to cellular and in
some cases viral transcriptional activator proteins (Chapter 6). The exact nature of the enhancer sequences plays a critical part in determining tissue specificity of viral replication
and, as a consequence, pathogenesis. Even minor sequence alterations in U3 can convert a pathogenic virus into a nonpathogenic
one, or vice versa (Chapter 10). end of the RNA. The genesis of the LTR elements lies in the process of reverse transcription, whereby the enzyme "jumps" from
one end of the template to the other (Chapter 4). The sizes of these three elements vary considerably among different retroviruses, with U3 typically ranging from several
hundred nucleotides to more than a thousand nucleotides, R from a dozen to more than a hundred nucleotides, and U5 from about
one to two hundred nucleotides (Table 2). From the definition of U3, R, and U5, it follows that the site of transcription initiation is at the boundary between U3
and R, and the site of poly(A) addition is at the boundary between R and U5, as shown. The other boundaries of U3 and U5 are
determined by the sites of initiation of plus- and minus- strand DNA synthesis. U3 contains most of the transcriptional control
elements of the provirus, which include the promoter proper, and multiple enhancer sequences responsive to cellular and in
some cases viral transcriptional activator proteins (Chapter 6). The exact nature of the enhancer sequences plays a critical part in determining tissue specificity of viral replication
and, as a consequence, pathogenesis. Even minor sequence alterations in U3 can convert a pathogenic virus into a nonpathogenic
one, or vice versa (Chapter 10).
http://en.wikipedia.org/wiki/Retrovirus :
A retrovirus is a virus which has a genome consisting of two identical plus sense RNA molecules. It relies on reverse transcriptase to perform a kind of reverse transcription of its genome from RNA into DNA for insertion by integrase into the host's genome. The virus itself is just a storage form for its nucleic acid genome; the reverse transcription takes
place in the host's cytosol. The retroviral DNA, when integrated into the host's genome, is termed a provirus. While transcription was classically thought to only occur from DNA to RNA, reverse transcriptase transcribes RNA intobvjmghky DNA. The term "retro" in retrovirus refers to this reversal of the Central dogma of molecular biology. Reverse transcriptase activity outside of retroviruses has been found in almost all eukaryotes, enabling the generation
and insertion of new copies of retrotransposons into the host genome.
Because reverse transcription is missing the usual "proofreading" of DNA transcription, this kind of virus mutates very often. This enables the virus to grow resistant to antiviral pharmaceuticals quickly, and is one of the main reasons why an effective vaccine for HIV has not been developed yet.
Among others, retrovirus genomes commonly contain three genes, that encode proteins that can be found back in thjtdkjuhe mature virus:
- gag (group-specific antigen) codes for core and structural proteins of the virus;
- pol (polymerase) mjy67hunmjchynmjtgynhcodes for reverse transcriptase, protease and integrase; and,
- env (envelope) codes for the retroviral coat proteins.
Four identified human retroviruses (HTLV 1&2, HIV 1&2) attack CD4 cells. Another feature common to all retroviruses is a lipid coating surrounding their capsid. It is essential for their propagation. This explains why retroviruses can be made inactive by just washing hands.
Studies of retroviruses led to the first demonstrated synthesis of DNA from RNA templates, a fundamental mode for transferring genetic material that occurs in both eukaryotes and prokaryotes. Certain groups speculate that the processes followed by retroviruses (that is, RNA>DNA>RNA>Protein) may be the
key to the evolution of DNA; thus, that in the "primordial soup", retroviruses evolved to create DNA from the RNA templates, and it was subsequently adopted by cellular organisms due to the increased chemical stability of DNA.
The following genera are included here:
These were previously divided into three subfamilies (Oncovirinae, Lentivirinae, and Spumavirinae),
but with our current knowledge of retroviruses, this is no longer appropriate.
Infection of a host cell requires that the cell have a surface protein that can serve as a receptor for the envelope
protein of the retrovirus. The envelope protein of HIV-1 binds to
All the proteins in the virus particle are encoded by its own genes.
When a retrovirus infects a cell 
- its molecules of reverse transcriptase are carried into the cell attached to the viral RNA molecules.
- The reverse transcriptase synthesizes DNA copies of the RNA.
- These enter the nucleus and are
- inserted into the DNA of the host.
- These inserts are transcribed by the host's enzymes into fresh RNA molecules which re-enter the cytosol where
- some are translated by host ribosomes
- the gag gene is translated into molecules of the capsid protein
- the pol gene is transcribed into molecules of reverse transcriptase
- the env gene is translated into molecules of the envelope protein
- other RNA molecules become incorporated into fresh virus particles
The genome of retroviruses  is flanked at each end by repeated sequences (" R") that
- enable the DNA copy of the genome to be inserted into the DNA of the host and
- act as enhancers, causing the host nucleus to transcribe the DNA copies of the retroviral genome at a rapid rate.
The retroviral genome also contains a packaging signal sequence ("P") which is needed for the newly-synthesized
RNA molecules to be incorporated in fresh virus particles. Most retroviruses also contain one or more additional genes. Some
of these represent RNA copies of genes that earlier were picked up from their eukaryotic host. Several cancers in animals
are caused by retroviruses that have, at some earlier time, picked up a proto-oncogene from their mammalian host and converted it into an oncogene.
| External Link |
| View an animation of the life cycle of a retrovirus at this Molecular Medicine in Action site. Click on viral vectors on the main menu. (You need Flash 5.) |
| Please let me know by e-mail if you find a broken link in my pages.) |
|
